When I first started my factory in China, I quickly learned that producing high-quality silicon carbide wasn’t just about smelting. One day, a German grinding wheel manufacturer rejected our shipment because of iron contamination. That’s when I discovered the critical importance of acid and alkali washing for abrasive grains.
Acid and alkali washing removes harmful impurities like iron compounds, free silicon, and silicon dioxide from abrasive grains. This chemical treatment not only purifies the material but also creates a rough surface texture that strengthens bonding with binders in grinding wheels, improving product quality and performance.
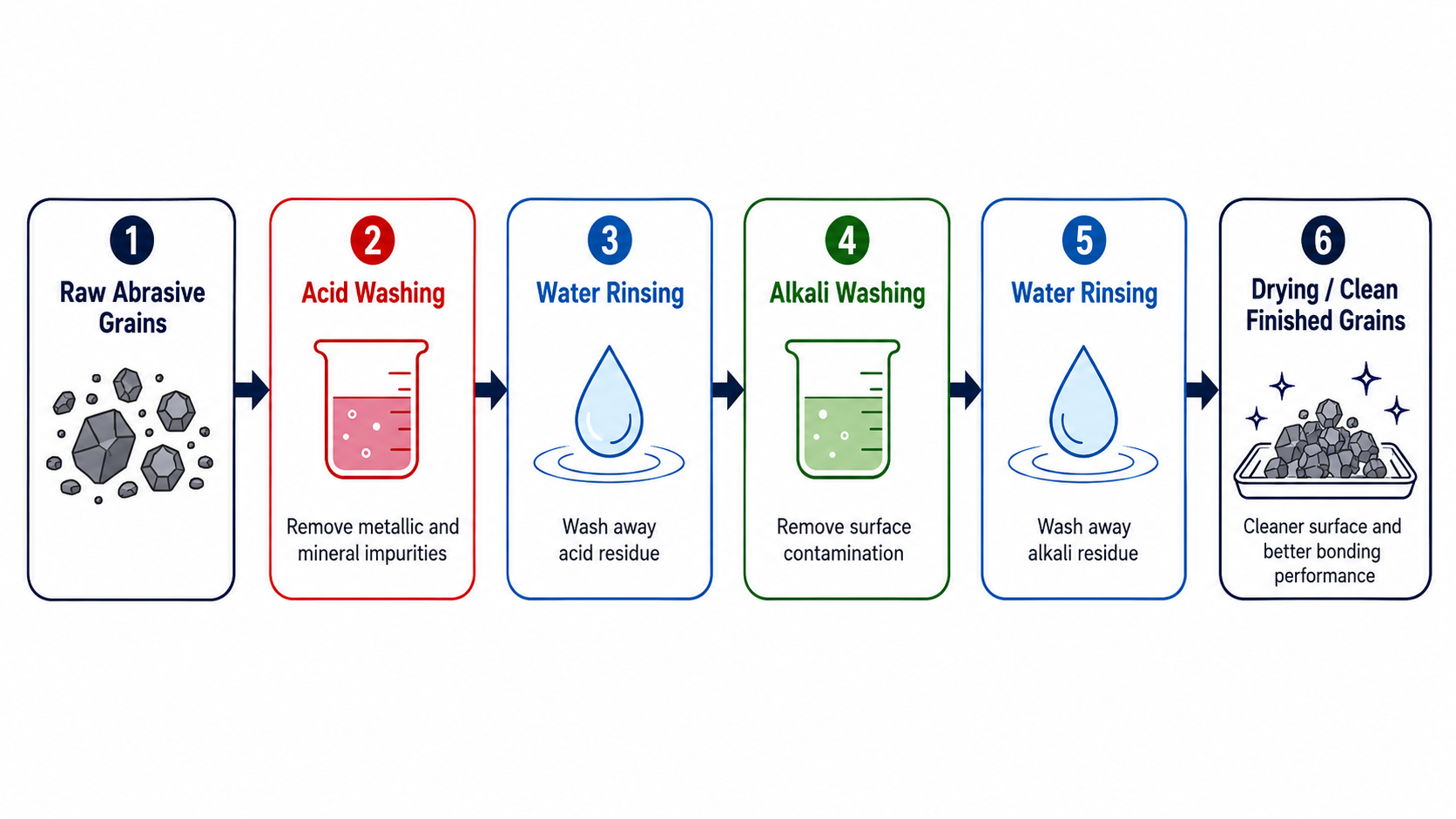
After that rejection, I spent months researching proper chemical treatment methods. I visited factories in Japan and studied their processes. Now, our products from Austria to the United States meet the strictest quality standards. Let me share what I’ve learned about why and how we use acid and alkali washing.
Why Do We Need Chemical Treatment for Abrasive Grains?
During my early production days, I thought smelting alone would purify the material enough. I was wrong, and it cost me dearly.
Chemical treatment removes impurities that magnetic separation cannot handle, including weakly magnetic and non-magnetic iron compounds, free silicon, and silicon dioxide. This process also roughens particle surfaces, increasing hydrophilicity and bonding strength with binders in abrasive tools.

The smelting process provides some purification. However, harmful impurities remain in the crystalline blocks. These include iron and its compounds, free silicon in silicon carbide, and silicon dioxide. Magnetic separation removes most strongly magnetic iron compounds. But it struggles with weakly magnetic or non-magnetic iron compounds.
Chemical treatment serves two main purposes. First, it removes these stubborn impurities through chemical reactions. Second, it modifies the particle surface. When acids and alkalis dissolve surface impurities, they create an uneven, rough texture. This roughness increases the particle’s water affinity and specific surface area. The result is stronger bonding between abrasive grains and binders in grinding wheels. This leads to higher tool strength and better performance.
What Makes Alkali Washing Essential for Silicon Carbide?
I remember when a French customer complained about discolored silicon carbide. The issue was free silicon and silicon dioxide on the surface. That’s when I understood alkali washing’s true value.
Alkali washing removes free silicon and silicon dioxide from silicon carbide surfaces. These impurities create chaotic “oxidation colors” and reduce hardness and grinding performance. The process uses sodium hydroxide, which reacts with these impurities to form water-soluble salts.

Free silicon and silicon dioxide on silicon carbide particles cause multiple problems. They give the material a messy, discolored appearance with various oxidation colors. More importantly, these impurities lower the abrasive’s hardness. They also reduce grinding performance significantly.
Alkali washing primarily uses sodium hydroxide (NaOH). The chemical reactions are:
| Impurity | Reaction | Result |
| Free Silicon | Si + 2NaOH → Na₂SiO₃ + 2H₂ | Sodium silicate (water-soluble) |
| Silicon Dioxide | SiO₂ + 2NaOH → Na₂SiO₃ + H₂O | Sodium silicate (water-soluble) |
These reactions produce sodium silicate, which dissolves in water. During rinsing, these salts wash away completely. The process also removes dust and graphite particles. This is why factories that perform chemical treatment usually skip separate water washing steps.
Corundum abrasives rarely need alkali washing. Free silicon and silicon dioxide are not their main impurities. This makes alkali washing a silicon carbide specialty process. In my factory, we alkali wash all silicon carbide products, especially those for coated abrasive manufacturers in Japan and the United States.
How Does Acid Washing Remove Iron Contamination?
Acid washing removes metallic iron, iron oxide, and other iron compounds from abrasive grains. These iron impurities cause spots on grinding wheels, affecting appearance and grinding quality. The process uses acids that convert iron compounds into water-soluble salts.

Iron contamination causes serious problems in abrasive products. When iron compounds remain on grain surfaces, they create visible spots on grinding wheels. These spots ruin the product’s appearance. More critically, they affect grinding quality and wheel performance. Customers in Germany and Austria, who demand the highest standards, will reject products with any iron contamination.
Acid washing uses various acids to remove iron impurities. The most common reactions include:
| Iron Compound | Acid Used | Reaction | Product |
| Metallic Iron | Hydrochloric acid | Fe + 2HCl → FeCl₂ + H₂ | Ferrous chloride (water-soluble) |
| Iron Oxide | Hydrochloric acid | Fe₂O₃ + 6HCl → 2FeCl₃ + 3H₂O | Ferric chloride (water-soluble) |
| Iron Oxide | Sulfuric acid | Fe₂O₃ + 3H₂SO₄ → Fe₂(SO₄)₃ + 3H₂O | Ferric sulfate (water-soluble) |
All these reactions produce water-soluble iron salts. These salts wash away easily during rinsing. The water also removes remaining dust, graphite, and other loose particles. This is why chemical treatment often replaces separate washing steps in production.
In my experience, proper acid washing is the difference between mediocre and excellent abrasive products. Our customers in the grinding wheel and coated abrasive industries can immediately tell the difference.
Which Grain Sizes Require Chemical Treatment?
When I started production, I chemically treated every grain size. My partner laughed and showed me I was wasting money. He taught me the practical approach that I still use today.
For silicon carbide, grains 150 mesh and finer must receive acid and alkali washing to produce qualified products. Coarser grains have varying requirements based on cost and quality targets. Brown aluminum oxide typically requires acid washing from 150 mesh, while white aluminum oxide starts from 70 mesh.
The grain size range for chemical treatment varies significantly between factories. Each producer balances cost against quality requirements. I’ve learned from visiting facilities that there’s no single “correct” answer. However, certain patterns exist.
Silicon Carbide Treatment Standards
For silicon carbide abrasives, industry practice shows clear patterns:
| Grain Size | Treatment Required | Reason |
| 150 mesh and finer | Acid and alkali washing mandatory | Cannot produce qualified products without treatment |
| Coarser than 150 | Varies by factory | Based on cost-quality balance |
For grains coarser than 150 mesh, I’ve observed three approaches in different factories:
Full Chemical Treatment Approach: Some factories perform both acid and alkali washing on all grain sizes. This produces the highest purity products. The cost is higher, but product quality is consistently excellent. Our factory uses this method for premium customers in Germany and France.
Alkali Washing Only: Other factories perform only alkali washing on coarser grains. This reduces cost while still removing free silicon and silicon dioxide. The purity is slightly lower than full treatment. However, it meets most standard requirements for grinding wheels and coated abrasives.
Water Washing Only: Some producers use only water washing for coarser grains. This has the lowest production cost. The silicon carbide appears darker in color. During washing, grain loss is minimal. However, product purity is lower. This approach struggles to meet premium product requirements.
The economic comparison is interesting. Water washing saves money initially. However, chemical treatment reduces losses during magnetic separation. Chemically treated grains have fewer iron impurities. This means less material gets pulled out during magnetic separation. The small amount of iron-containing material removed can be reprocessed through acid washing.
I recommend discarding accumulated iron-containing material every two to three months. After several cycles, these grains accumulate too much iron for effective reprocessing. Overall, when considering the entire production process, chemical washing and water washing yield similar final product rates. The real difference is purity level and customer acceptance.
Corundum Treatment Requirements
Corundum abrasives have different treatment needs than silicon carbide. I learned this when I expanded from silicon carbide to aluminum oxide production.
| Abrasive Type | Starting Mesh for Acid Washing | Alkali Washing |
| Brown Aluminum Oxide | 150 mesh | Generally not required |
| White Aluminum Oxide | 70 mesh | Generally not required |
Brown aluminum oxide typically starts acid washing from 150 mesh. White aluminum oxide requires earlier treatment, often starting from 70 mesh or even coarser. This is because white aluminum oxide has stricter purity requirements. Customers using white aluminum oxide, especially in precision grinding applications, demand extremely low iron content.
Some factories use calcination instead of acid washing for corundum. Calcination is a heat treatment process. It removes certain impurities through high-temperature oxidation. This method works well for some applications. However, acid washing remains more effective for removing iron compounds.
Alkali washing is rarely necessary for corundum abrasives. Free silicon and silicon dioxide are not major impurities in aluminum oxide materials. The crystal structure of corundum naturally resists these contaminants. This is a significant cost advantage compared to silicon carbide production.
In my factory, we follow these industry standards but adjust based on customer requirements. Our grinding wheel customers in Austria prefer the highest purity grades. For them, we often start chemical treatment at coarser grain sizes than standard practice. Our distributor customers in the United States accept standard treatment ranges. This flexibility helps us maintain competitive pricing while meeting diverse quality needs.
The key lesson I’ve learned is that chemical treatment requirements depend on three factors: grain size, abrasive type, and final application requirements. Understanding these factors helps balance cost and quality effectively. This knowledge has been crucial for our success in international markets.
Conclusion
Chemical treatment through acid and alkali washing is essential for producing high-quality abrasive grains. It removes harmful impurities, improves bonding strength, and ensures consistent performance in grinding wheels and coated abrasives. The specific treatment requirements vary by abrasive type and grain size, allowing producers to balance cost and quality based on customer needs.





